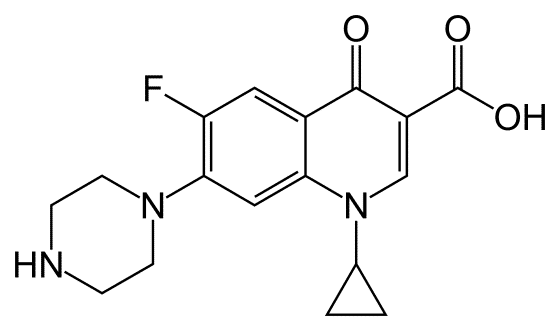
Following previous work on the potential risk of acute liver failure associated with quinolone antibiotics, RSI investigators, Mohamed Taher, Franco Momoli, Donald Mattison and Daniel Krewski evaluated the potential risk of retinal detachment (RD) associated with these medications.
Although no cases of RD were linked to quinolones in clinical trials, some reports of RD were noted in the US FDA Adverse Event Reporting System in conjunction with systemic use of quinolone antibiotics. Analysis of electronic health records from over 500 US healthcare institutions revealed elevated but non-significant risks in African Americans (ciprofloxacin and levofloxacin), those aged 56–70 years old (moxifloxacin), and women (ciprofloxacin).
The authors noted that these suggestions of increased risk observed in some population subgroups warrant further investigation.
Experts related to this news item
More RSI News
RSI chairs panel session at Building the Next Generation of Canadian Infrastructure Conference
RSI chairs and presents on panel sessions for the Toronto conference, “Building the Next Generation of Canadian Infrastructure: Defining a roadmap for optimizing infrastructure planning,…
RSI Chairs Joint FAO/WHO Expert Meeting on Microbiological Risk Assessment (JEMRA)
RSI’s Dr. Emma Hartnett chaired the recent Joint FAO/WHO Expert Meeting on Microbiological Risk Assessment (JEMRA). The focus of the week-long event held March 11…
RSI partners with Engineers Canada to offer Online Workshop on the PIEVC Protocol
RSI and Engineers Canada are partnering to offer an online version of the introductory workshop on the PIEVC Protocol for infrastructure climate risk and vulnerability…
RSI contributes to newly published book on Blood Safety.
Written by experts in the field, Blood Safety: A Guide to Monitoring and Responding to Potential New Threats offers a concise yet comprehensive overview on critical issues in monitoring…



