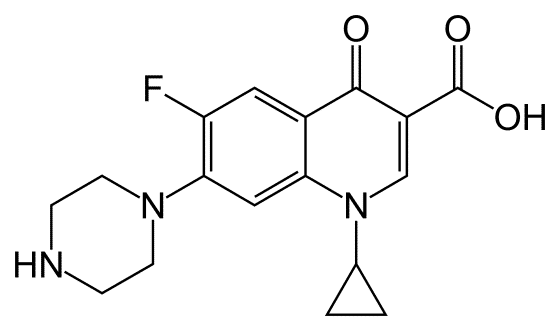
Following previous work on the potential risk of acute liver failure associated with quinolone antibiotics, RSI investigators, Mohamed Taher, Franco Momoli, Donald Mattison and Daniel Krewski evaluated the potential risk of retinal detachment (RD) associated with these medications.
Although no cases of RD were linked to quinolones in clinical trials, some reports of RD were noted in the US FDA Adverse Event Reporting System in conjunction with systemic use of quinolone antibiotics. Analysis of electronic health records from over 500 US healthcare institutions revealed elevated but non-significant risks in African Americans (ciprofloxacin and levofloxacin), those aged 56–70 years old (moxifloxacin), and women (ciprofloxacin).
The authors noted that these suggestions of increased risk observed in some population subgroups warrant further investigation.
Experts related to this news item
More RSI News
Willhite receives prestigious SOT award
Calvin C. Willhite, an Associate Expert with Risk Sciences International working on a range of toxicological risk issues, was just announced as the recipient of the prestigious…
RSI gives keynote
RSI experts Jennifer Go and Daniel Krewski, were recently invited to present a paper on maternal and child environmental heath during a global, virtual workshop originating in Foshan, Guangdong, China.
RSI hosts Mitacs intern
Risk Sciences International will host Abdallah Alami as an intern in statistics with support from the Mitacs Accelerate program. This unique university-industry program supports interns at…
Dr. Mattison chairs NAM report
Compounded medications prepared by pharmacists or physicians have been used extensively throughout history as a mainstay of therapeutics. Over the past half-century – with the…



