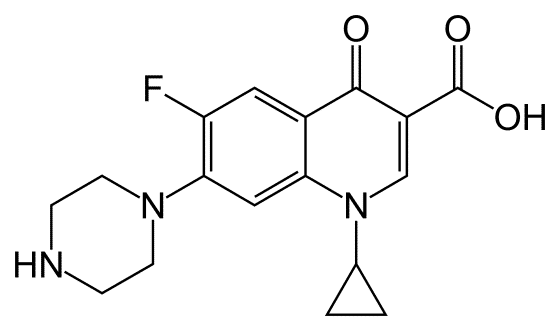
Following previous work on the potential risk of acute liver failure associated with quinolone antibiotics, RSI investigators, Mohamed Taher, Franco Momoli, Donald Mattison and Daniel Krewski evaluated the potential risk of retinal detachment (RD) associated with these medications.
Although no cases of RD were linked to quinolones in clinical trials, some reports of RD were noted in the US FDA Adverse Event Reporting System in conjunction with systemic use of quinolone antibiotics. Analysis of electronic health records from over 500 US healthcare institutions revealed elevated but non-significant risks in African Americans (ciprofloxacin and levofloxacin), those aged 56–70 years old (moxifloxacin), and women (ciprofloxacin).
The authors noted that these suggestions of increased risk observed in some population subgroups warrant further investigation.
Experts related to this news item
More RSI News
Country Foods Forum
Leading up to February 11 and 12, 2020, Risk Sciences International had the privilege of assisting Health Canada in its organization of the Country Foods…
Workshop on Evidence Integration December 5-6, 2019
Risk Sciences International is organizing a two day Workshop on Evidence Integration in support of human health risk assessment in collaboration with the McLaughlin Centre…
RSI wins 2019 Consulting Engineers of Ontario Award of Merit
Risk Sciences International (RSI), as members of a consulting team with Stantec, along with the Ontario First Nations Technical Services Corporation (OFNTSC), has won an Award of Merit from the…
Updated CSA Technical Guide for Infrastructure in Permafrost published
A second updated edition of the CSA national permafrost standard was published on May 21, 2019 and features climate and climate change information provided by…



