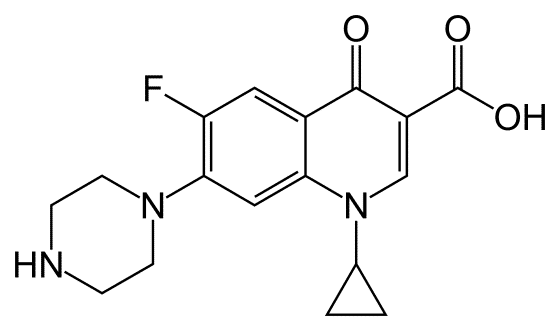
Following previous work on the potential risk of acute liver failure associated with quinolone antibiotics, RSI investigators, Mohamed Taher, Franco Momoli, Donald Mattison and Daniel Krewski evaluated the potential risk of retinal detachment (RD) associated with these medications.
Although no cases of RD were linked to quinolones in clinical trials, some reports of RD were noted in the US FDA Adverse Event Reporting System in conjunction with systemic use of quinolone antibiotics. Analysis of electronic health records from over 500 US healthcare institutions revealed elevated but non-significant risks in African Americans (ciprofloxacin and levofloxacin), those aged 56–70 years old (moxifloxacin), and women (ciprofloxacin).
The authors noted that these suggestions of increased risk observed in some population subgroups warrant further investigation.
Experts related to this news item
More RSI News
RSI Provides Technical Advice and Review to Vietnam Partners and on-site Climate Support for Workshop
Engineers Canada is a partner with the German development agency Deutsche Gesellschaft für Internationale Zusammenarbeit GmbH (GIZ) in the execution of their Enhanced Climate Services…
Neil Comer to present at OPTrust Climate Change Symposium
The OPTrust Climate Change Symposium being held NOVEMBER 19-20, 2018 in TORONTO offers an opportunity to provide new insights into how to face climate change…
Emma Hartnett 2018 recipient of Center for Produce Safety (CPS) Research Award
CPS has recently announced 13 new research awards totaling a little over $2.6 million. The awards are for research projects directed at answering critical questions…
Best Practices for Consideration of the Effects of Climate Change in Project-Level Assessments
Risk Sciences International (RSI) is pleased to announce in collaboration with the Ontario Centre for Climate Impacts and Adaptation Resources (OCCIAR), the public release of…



