Client: Health Canada
Listing of the client in no way affirms the client's support, sponsorship, or validation in any form of Risk Sciences International or the RSI staff member(s) who conducted this project during their stay with RSI or prior to joining the company. This case study is displayed for informative purposes only to demonstrate the capacity of RSI staff members. This case study reveals no proprietary information or information deemed sensitive.
Finalization of Risk Assessment on Secure Opioid Dispensing Devices
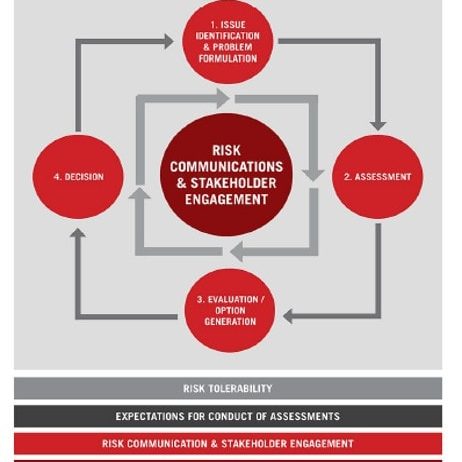
To support decision-making around modifications to opioid agonist treatment delivery, a detailed risk assessment was requested to evaluate the integration of secure dispensing devices for direct patient access to hydromorphone. Building on a previously conducted preliminary assessment, the work required a structured framework that would articulate hazards, assess likelihoods and risks, and identify mitigation strategies associated with introducing these devices into service delivery models.
The client sought to understand the public health and safety implications of transitioning from conventional in-person opioid dispensing to a model incorporating automated secure devices. This included analyzing risks to intended and unintended users, security risks such as theft or misuse, and any system vulnerabilities in the dispensing, recording, and delivery processes. RSI was tasked with reviewing technical documentation, examining standard operating procedures (SOPs), and conducting key informant interviews with both the device manufacturer and provincial project proponents. The aim was to validate and update the risk profile in light of new operational details and anticipated public safety impacts.
The final output included an updated risk assessment report and a formal presentation of findings. These deliverables were intended to assist Health Canada’s Controlled Substances Directorate in evaluating whether the proposed devices offered a net benefit in reducing harm among populations vulnerable to toxic illicit drug supplies.
Experts related to this case study
More RSI Case Studies
RSI presents a very small selection of case studies to highlight some of its key work.