Client: Health Canada
Listing of the client in no way affirms the client's support, sponsorship, or validation in any form of Risk Sciences International or the RSI staff member(s) who conducted this project during their stay with RSI or prior to joining the company. This case study is displayed for informative purposes only to demonstrate the capacity of RSI staff members. This case study reveals no proprietary information or information deemed sensitive.
Evaluation of Drug Recall Communication and Risk Determination Processes
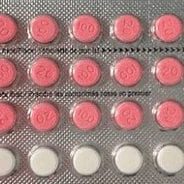
In response to public and ministerial concerns about the timeliness and adequacy of information shared during a major prescription drug recall, Health Canada commissioned an independent review to assess its response to the event. The situation involved a recall of the oral contraceptive Alysena, issued due to packaging issues that altered the intended dosing sequence and raised questions about potential unintended health risks.
Health Canada sought a comprehensive evaluation of the processes surrounding the recall, with particular emphasis on regulatory responsibilities, risk characterization, service standard adherence, and communication effectiveness. Specifically, the department required a reconstruction of the recall timeline, an analysis of the roles and legal mandates of both Health Canada and the manufacturer, and an assessment of the communications issued internally, with external stakeholders, and to the public.
The review was expected to determine whether Health Canada’s actions—especially related to risk evaluation and public notifications—were consistent with its existing authorities and service expectations. Additionally, the evaluation explored whether differing risk assessments between the regulator and the manufacturer may have influenced the timing and clarity of the recall messaging. Broader reflections on statutory limitations and opportunities to strengthen recall protocols were also included.
Consultations with departmental staff and access to internal documentation informed the final report, which was delivered alongside a presentation to senior management to support ongoing improvements in drug safety oversight and public health protection.
Experts related to this case study
More RSI Case Studies
RSI presents a very small selection of case studies to highlight some of its key work.