Mandate
In 2013, due to public concern and criticism of Health Canada’s apparently delayed response to a drug company recall of an oral contraceptive, the then Minister of Health initiated an investigation to determine if the relevant processes were followed and if they were sufficient. RSI was engaged to conduct this review.
The concern arose when generic drug maker Apotex notified its wholesalers and retailers that it had initiated a Type II recall of a single lot of AlysenaTM 28, because one package of the drug with an incorrect proportion of active and placebo pills had been discovered. Due to a confluence of several factors, members of the public learned of the recall several days before Health Canada posted the recall notice on its website. Widespread media and public criticism charged that Health Canada had not responded with appropriate speed and concern, and showed a lack of respect for women’s health. Many of the critical claims were based on misunderstandings of the number of faulty packages that had been found and that may have been released to the Canadian market, the sequence of events that led to the recall announcement, and of the extent of Health Canada’s powers to recall a faulty drug. Nevertheless, the competence of Health Canada’s drug safety processes was called into question and the review of the process was initiated.
Work
The review involved an analysis of the events and processes that led to public criticism of Health Canada’s handling of the drug company’s recall of a contraceptive drug due to packaging errors. The evaluation consisted of several components.
First, a review and analysis were conducted of the regulatory and procedural basis of the department’s roles responsibilities and powers as set out in the relevant legislation and regulations, and policy and guidance documents. Second, key individuals who had been involved in the response to the recall, or were in roles with related responsibilities, were interviewed. Third, through the interviews and external materials, a chronology of the events of the recall and the departmental response was assembled as an aid to evaluating the appropriateness of the response. Finally, a review of the media depiction and public response to the recall was conducted, to understand the way that the events were interpreted, and were expressed as concerns within the public.
The analysis considered the events that contributed to the circumstances of the recall, an evaluation of the recall events and the wider response; and a review of Health Canada’s handling of the recall.
Outcomes
RSI’s analysis of the events, and evaluation of Health Canada’s response, led to the development of a set of recommendations under four main themes. Three of these related to internal Health Canada processes: they were that Health Canada should improve its capacity to respond to company recall notifications in a timely manner; that roles and responsibilities related to recall decisions and response be clarified and communicated; and that in future recall decisions, Health Canada should include a consideration of the relevant levels of social concern. The final recommendation concerned higher-level government arrangements, suggesting that any future updates of the relevant legislation should include improved and clarified drug company notification requirements.
Posted in Case Study
More RSI Case Studies
RSI presents a very small selection of case studies to highlight some of its key work.
The Alliance of Blood Operators (ABO) needed a coherent risk management framework to guide decision-making on the delivery of blood services by its member organizations, which would help produce consistent decision-making processes and outcomes across the different national jurisdictional and regulatory structures. The RBDM approach was meant to support a shift in risk decision-making in national blood services organizations from one focused on minimizing all risks to blood quality and supply, with little concern for costs, to a risk-based approach in which resources are allocated according to risk and the application of financial resources and management rigour is proportional to the risk they are addressing.
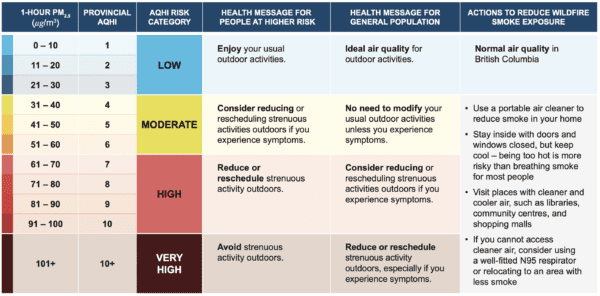
Read MoreEnvironment and Climate Change Canada, with Health Canada, wanted to understand perceptions of the health risks from smoke from wildfires and from residential burning (winter heating using wood stoves, and fireplaces), and public health communication approaches and messages used in these contexts. This information would support revisions of the health messages used in the Air Quality Health Index (AQHI). The AQHI expresses the health risk associated with three common urban air pollutants, and is calculated at monitoring stations in communities across Canada to inform the public of the risk level of current local air quality conditions.
Read MoreThe project began with a series of consultations with UN-Habitat at UNON in Nairobi. In addition to Cemil Alyanak and Nicholas you, the project grew to include many key stakeholders both within UN-Habitat and from other organizations. Among the key organizations included in the early stages were United Cities and Local Governments, Cities Alliance, the World Bank, Habitat for Humanity, Cisco, and UN agencies interested in urban health, notably the World Health Organization.
Read MoreUnder the supervision of Director Purcell and his cabinet, the six-month project began with an internal review of attitudes, projects, responsibilities and management reporting lines. The work uncovered multiple inconsistencies and inefficiencies. What’s more, there were very real risk consequences at the operational level.
Read MoreThe original mandate aimed to find a new direction for the world renowned International Herald Tribune just as both the World Wide Web was in its infancy, and the newspaper faced increased competition from international and regional editions of established newspapers and magazines such as The Economist, The Wall Street Journal, and The Financial Times.
Read More