In its 2007 report on Toxicity Testing in the 21st Century: A Vision and a Strategy, the US National Research Council (NRC) highlighted need for more rapid toxicity testing strategies in order to expand the coverage of the large number of chemicals for which toxicological data is lacking.
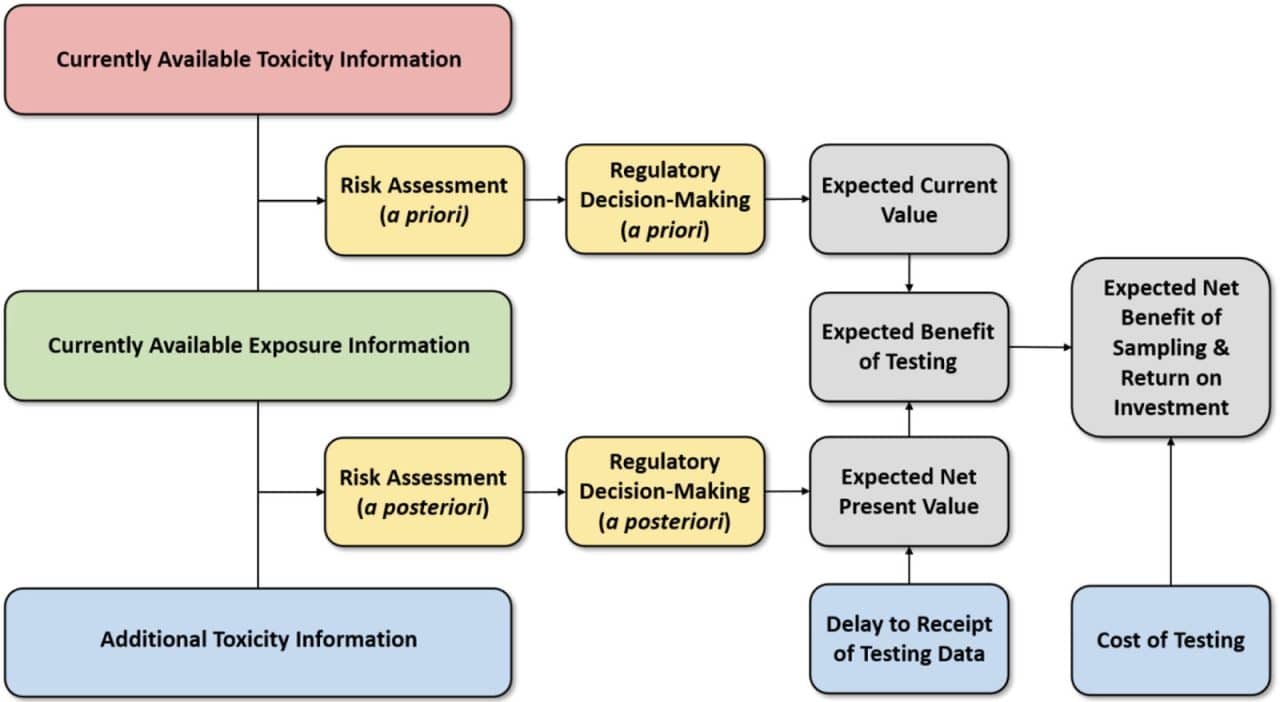
This need was further underscored by the NRC in its 2009 report Science and Decision: Advancing Risk Assessment, which recommended the use of formal value-of-information (VOI) techniques to evaluate alternative testing strategies. Hagiwara et al. (2022) recently developed VOI methods to evaluate the trade-offs between the cost, timeliness, and uncertainty reduction in toxicity testing.
These contributions suggest that while rapid alternative test methods may be subject to greater uncertainty than traditional more expensive animal tests of longer duration, having sufficient information to support risk decision making in a timely manner can result in a lower total social cost by avoiding pubic health impacts that can accrue when decisions are delayed pending the results of toxicity tests of longer duration. Although further VOI analyses are needed to reaffirm these findings under broader real-world toxicity testing scenarios, initial results appear to be supportive of alternative test methods that can provide cost-effective toxicity data in a timely manner.
More RSI News
RSI gives keynote
RSI experts Jennifer Go and Daniel Krewski, were recently invited to present a paper on maternal and child environmental heath during a global, virtual workshop originating in Foshan, Guangdong, China.
RSI hosts Mitacs intern
Risk Sciences International will host Abdallah Alami as an intern in statistics with support from the Mitacs Accelerate program. This unique university-industry program supports interns at…
Dr. Mattison chairs NAM report
Compounded medications prepared by pharmacists or physicians have been used extensively throughout history as a mainstay of therapeutics. Over the past half-century – with the…
Country Foods Forum
Leading up to February 11 and 12, 2020, Risk Sciences International had the privilege of assisting Health Canada in its organization of the Country Foods…
