In its 2007 report on Toxicity Testing in the 21st Century: A Vision and a Strategy, the US National Research Council (NRC) highlighted need for more rapid toxicity testing strategies in order to expand the coverage of the large number of chemicals for which toxicological data is lacking.
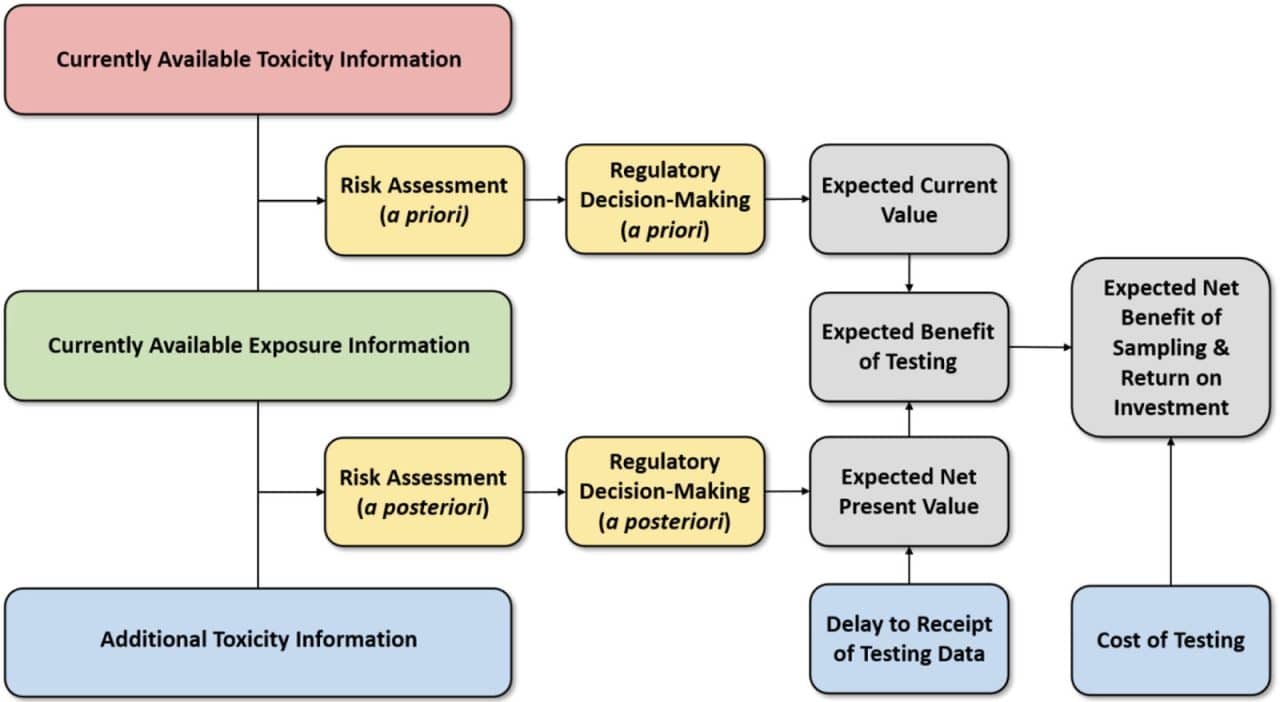
This need was further underscored by the NRC in its 2009 report Science and Decision: Advancing Risk Assessment, which recommended the use of formal value-of-information (VOI) techniques to evaluate alternative testing strategies. Hagiwara et al. (2022) recently developed VOI methods to evaluate the trade-offs between the cost, timeliness, and uncertainty reduction in toxicity testing.
These contributions suggest that while rapid alternative test methods may be subject to greater uncertainty than traditional more expensive animal tests of longer duration, having sufficient information to support risk decision making in a timely manner can result in a lower total social cost by avoiding pubic health impacts that can accrue when decisions are delayed pending the results of toxicity tests of longer duration. Although further VOI analyses are needed to reaffirm these findings under broader real-world toxicity testing scenarios, initial results appear to be supportive of alternative test methods that can provide cost-effective toxicity data in a timely manner.
More RSI News
Dr. Emma Hartnett Selected for JEMRA Roster of Scientific Experts
RSI’s Emma Hartnett has been selected to be part of the Joint FAO/WHO Expert Meetings on Microbiological Risk Assessment (JEMRA) Roster of Experts for a…
Safety Profiles of Newer Contraceptives presented at meeting of Society of Reproductive Investigation
RSI Chief Medical Officer, Dr. Donald R. Mattison, in collaboration with colleagues from the Institute for Safe Medication Practices and Wake Forest University, evaluated and…
Training Seminar in Systematic Review
RSI recently conducted an in-house training session in systematic review for its staff. The session featured a keynote presentation by Dr. Katya Tsaioun from Johns…
Decision making framework developed for Canadian Blood Services (CBS) now online
RSI was contracted by CBS to assist the Alliance of Blood Operators in the development of a blood-safety oriented risk-based decision-making framework. Why use this…
